TOP-10 of the Russian seaports
Compared to the corresponding period of 2019, the cargo turnover of the Russian seaports for January-September 2020 has reduced by 2,7% and amounted to 611,5 million tons. Under the global economic crisis and decline in global trade, the volume of cargo transshipment has decreased for the first time in 24 years.
Since 2000, the cargo turnover of the Russian seaports has increased 4 times, and the highest figure of 840,3 million tons was recorded last year (see Picture 1). For reference, the peak of cargo turnover of all the USSR ports was recorded in 1989 and amounted to 407 million tons.
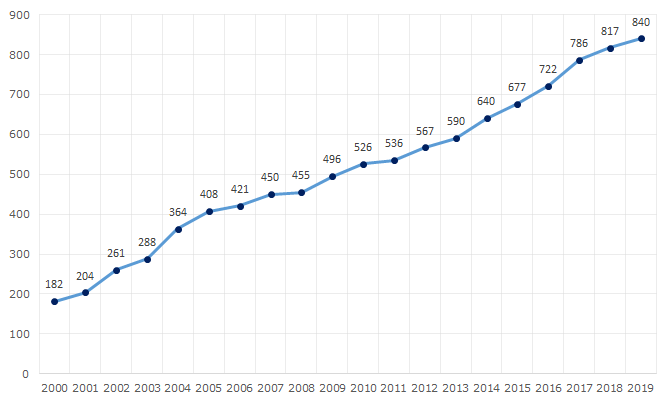 Picture 1. Dynamics of the cargo turnover of the Russian seaports, million tons
Picture 1. Dynamics of the cargo turnover of the Russian seaports, million tonsCurrently, there are 67 seaports in Russia. Novorossiysk Commercial Seaport is the largest domestic harbor with 156,8 million tons of the cargo turnover (see Table 1). The second one is Ust-Luga (Leningrad region) seaport handling over 100 million tons of cargo. All Russian goods servicing in the seaports of the Baltic States are forwarded to this port. Recently, the government of the Republic of Belarus declared about plans on forwarding all cargoes from Lithuanian and Latvian seaports to Ust-Luga.
For reference: the largest European seaport – Rotterdam (the Netherlands) – served 469 million tons of cargo in 2018. In 2017, China's Ningbo-Zhoushan, the largest port in the world, handled 1 077 million tons of cargo.
| Rank | Port | Cargo turnover in 2019, million tons | Share, % | Basin |
| 1 | Novorossiysk | 156,8 | 18,7% | Azov-Black Sea |
| 2 | Ust-Luga | 103,9 | 12,4% | Baltic |
| 3 | Vostochniy | 73,5 | 8,7% | Far Eastern |
| 4 | Murmansk | 61,9 | 7,4% | Arctic |
| 5 | Primorsk | 61,0 | 7,3% | Baltic |
| 6 | Big Port of Saint Petersburg | 59,8 | 7,1% | Baltic |
| 7 | Vanino | 31,4 | 3,7% | Far Eastern |
| 8 | Sabetta | 27,7 | 3,3% | Arctic |
| 9 | Nakhodka | 25,6 | 3,0% | Far Eastern |
| 10 | Tuapse | 25,2 | 3,0% | Azov-Black Sea |
| TOP-10 of seaports | 626,8 | 74,6% | - | |
| Total | 840,3 | 100 | - | |
Source: the Federal Agency for Maritime and River Transport, Association of Commercial Seaports; analysis by Credinform
654 million tons or 77,8%, i.e. the majority of cargoes handled in the seaports of Russia is exported (see Picture 2).
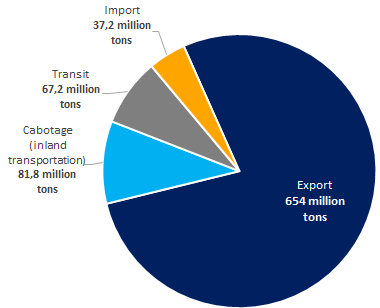 Picture 2. The structure of the cargo traffic in the seaports of Russia in 2019, million tons
Picture 2. The structure of the cargo traffic in the seaports of Russia in 2019, million tonsThe following handled goods prevail in the Russian seaports: crude oil – 276 million tons, coal – 176 million tons, petroleum products (gasoline, oils, fuel oil, etc.) – 150 million tons, breakbulk cargo – 57 million tons (see Picture 3).
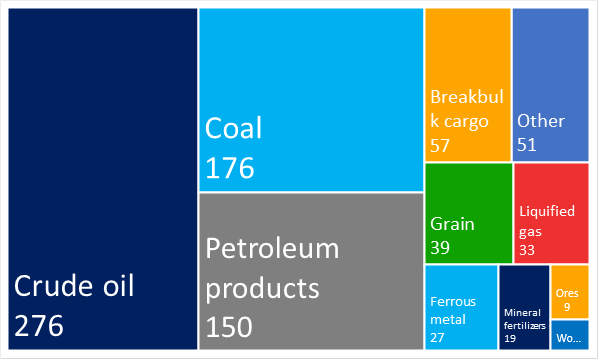 Picture 3. The structure of cargoes handled in the seaports of Russia in 2019, million tons
Picture 3. The structure of cargoes handled in the seaports of Russia in 2019, million tonsChanges in legislation
Starting from January 1st, 2021 the new procedure for licensing of the production and technical maintenance of medical equipment will come into force.
The corresponding Provision is approved by the Decree of the Government of the Russian Federation as of 15.09.2020 No. 1445.
The licensing authority for this type of activity is the Federal Service for Surveillance in Healthcare (Roszdravnadzor).
The license applicants are to meet the following obligatory requirements while operating:
- availability of appropriate facilities owned by property rights or on other legal basis in the place of activity;
- availability of the technical means and equipment, measuring tools with the manufacturer’s technical documents and corresponding requirements to its examination, stated in the article 13 of the Federal law “On ensuring the uniformity of measurements”;
- availability of registration certificates on medical equipment, confirming the production of this equipment or relation to the place of its manufacture;
- availability of agreements between the manufacturer and the license applicant on organization of manufacture, which should contain the criteria of quality and safety control assurance of the manufactured products, as well as responsibility to the third parties;
- availability of quality management system, in accordance with the requirements of international standard GOST ISO 13485-2017;
- availability of staff members, having labor contracts, responsible for the manufacture and quality of the medical equipment, with higher or secondary professional (technical) education, having the work experience for at least 3 years in this field and supplementary vocational education (advance training at least once in 5 years) in the field of work performed and services rendered.
The full list of licensing requirements for both license applicants and license holders is contained in the Appendix to this Decree of the Government.
The information relevant to these types of activities, provided by part 1 and 2 of the article 21 of the Federal law ‘On licensing certain activities’ is published in the official web-site of Roszdravnadzor no later than 10 days after the date of arrival at the decision on grant, re-issuance, suspension, renewal or termination of the license.
A state fee is charged for the issuance or renewal of license.
All the licenses issued earlier are to be reissued in accordance with the requirements of the new Decree until December 31st, 2023. This Decree is valid until January 1st, 2027.
The Provision as of 03.06.2013 №469 approved earlier by the Decree of the Government will be invalid since January 1st, 2021.
The users of the Information and Analytical system Globas have access to all the available information (including archive data) about entities engaged in production and technical maintenance of medical equipment. Currently, more than 5300 companies have licenses for these types of activities.